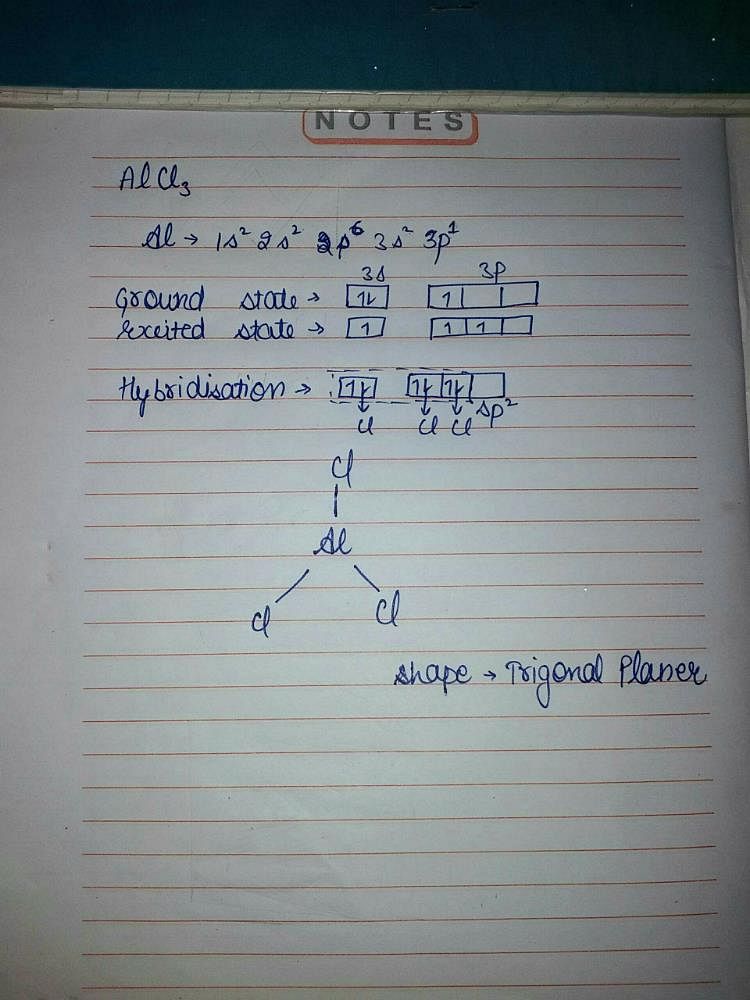
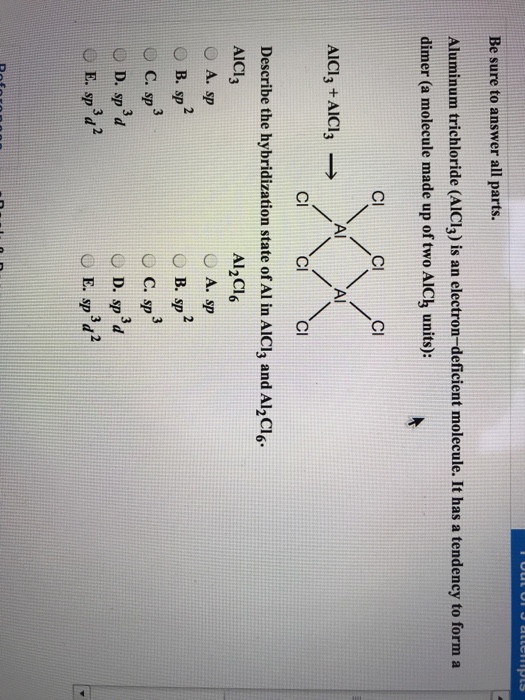
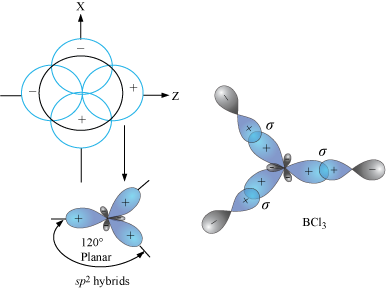
Describe the change in hybridisation (if any) of the `AI` atom in the following: `AlCl_(3)+Cl^(?... - YouTube

Mention the hybridization of the central atom in the given molecule. Molecular AlCl3 | Homework.Study.com
6. Which one of the following compounds has the electron pair geometry as the trigonal bipyramid with three equatorial positions occupied by lone pairs of electrons? A) [AlCl3] B) XeF2 C) [

AlCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, bond angle | Molecular geometry, Molecular, Molecular shapes

Account for the hybridization at each carbon atom in the mentioned molecule. Chloroprene(used to make neoprene, a synthetic rubber) | Homework.Study.com

SOLVED: Which molecule has a central atom that is sp3 hybridized? Select the correct answer below: SF6 CH3Cl AlCl3 PCl5

AlCl3 Lewis Structure, Molecular Structure, Hybridization, Bond Angle, and Shape - Geometry of Molecules