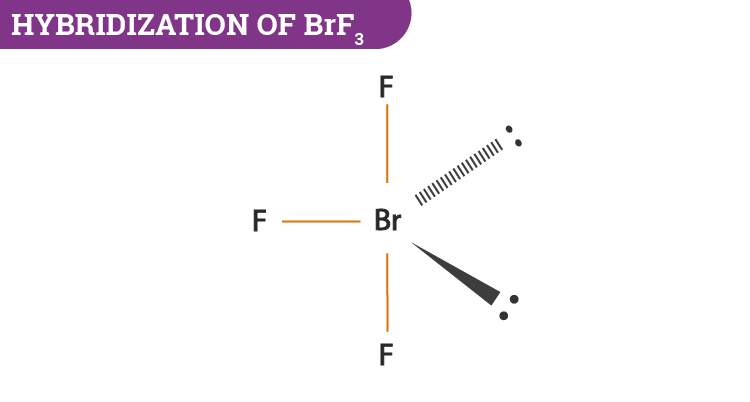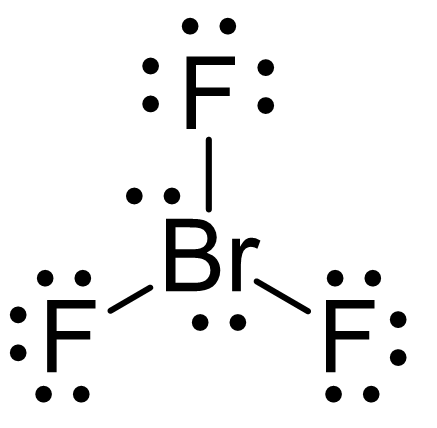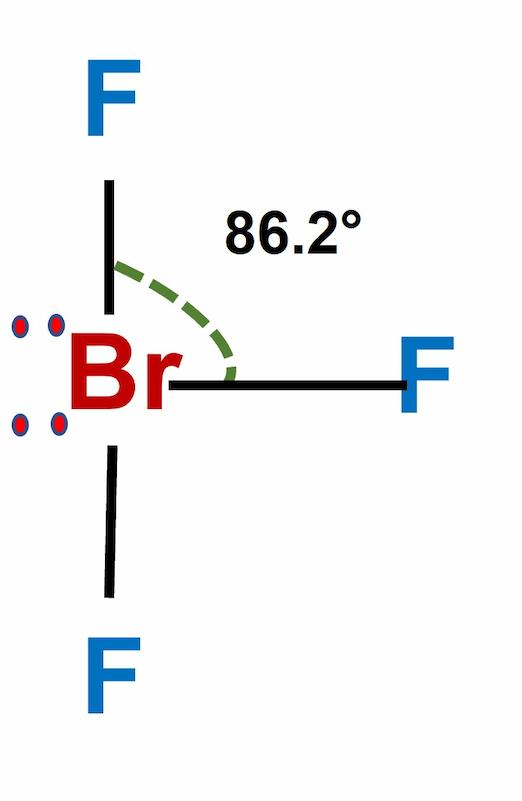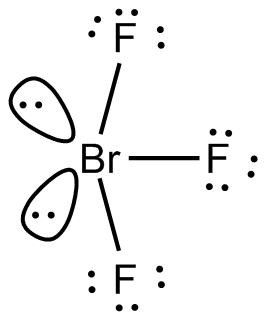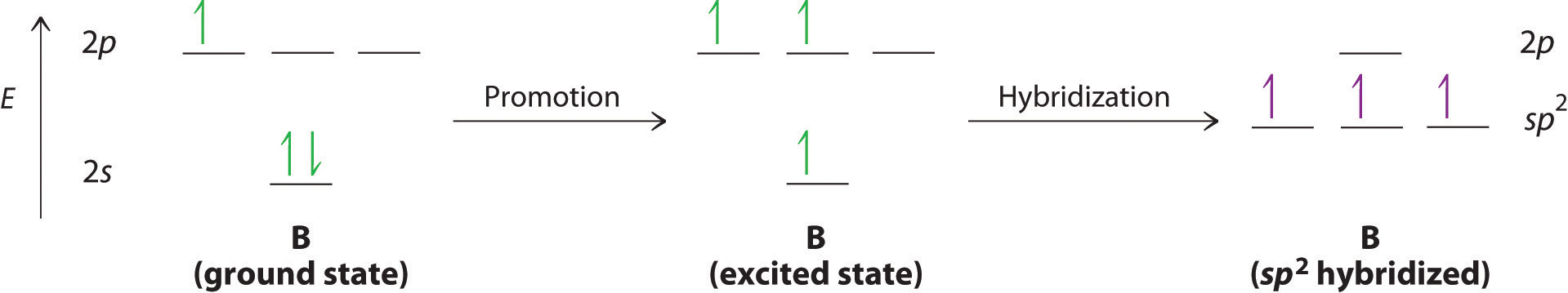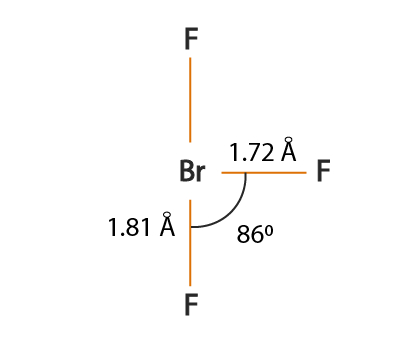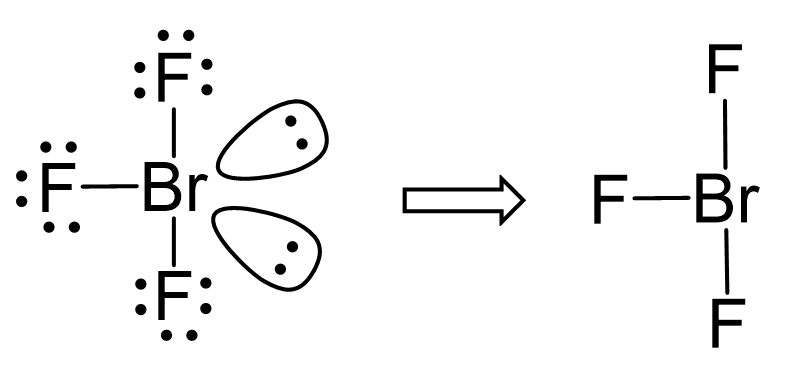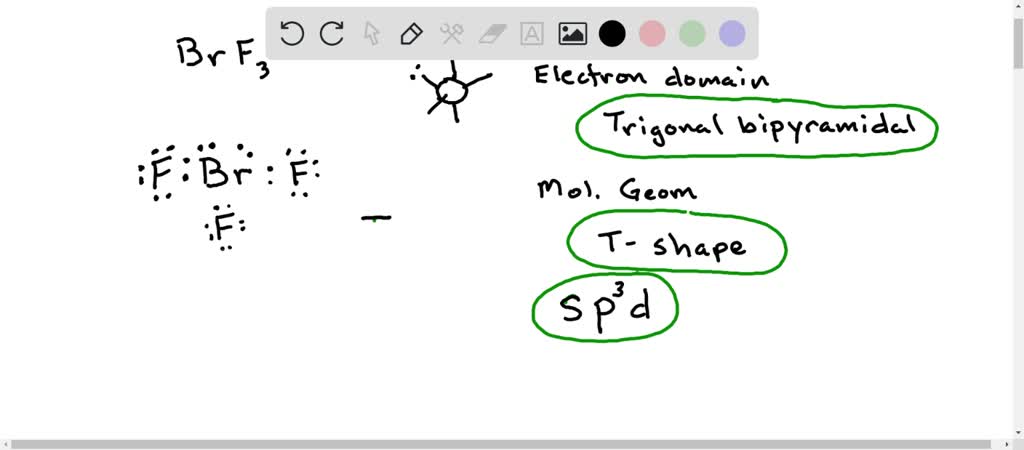
SOLVED: Consider the molecule BrF3. What is the electron domain geometry for this molecule? What is the molecular geometry for this molecule? What is the hybridization for this molecule? (answer in the

The hybridization and geometry of Br{F}_{3} molecules are:{ sp }^{ 3 }{ d }^{ 2 } and tetragonal{ sp }^{ 3 }d and T-shaped{ sp }^{ 3 }d and bentnone of these

BrF3 Lewis structure, molecular geometry or shape, electron geometry, bond angle, hybridization | Molecular geometry, Molecular, Electron configuration