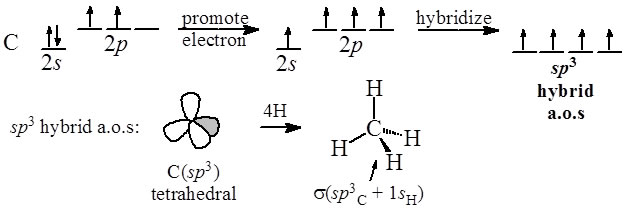
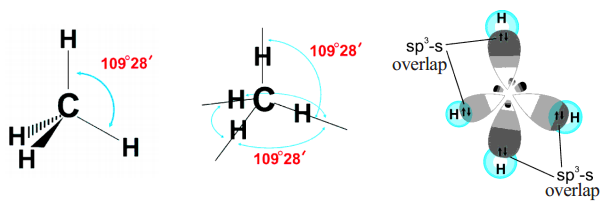
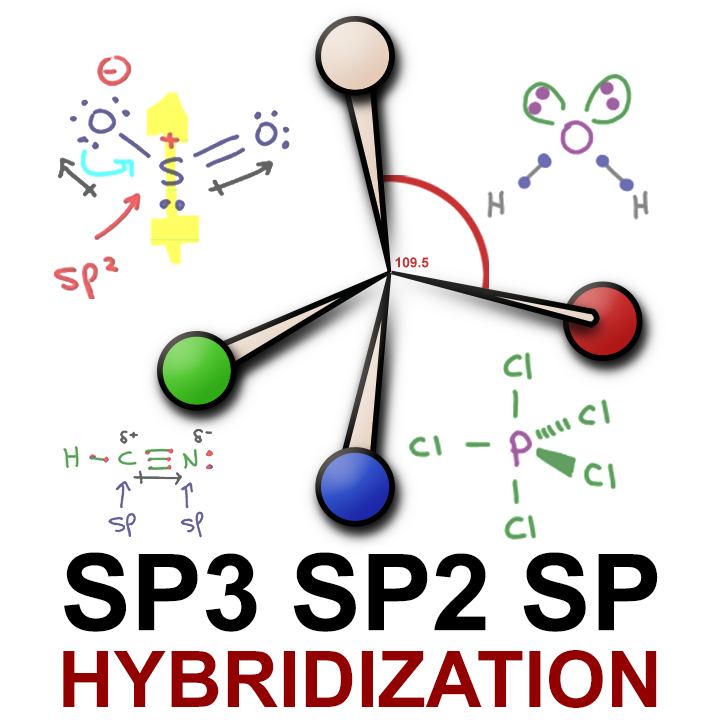
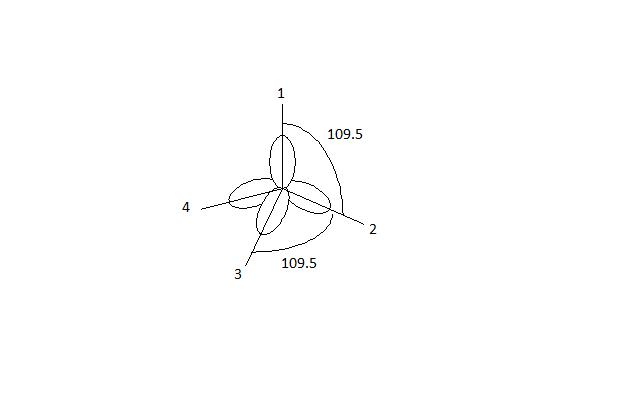
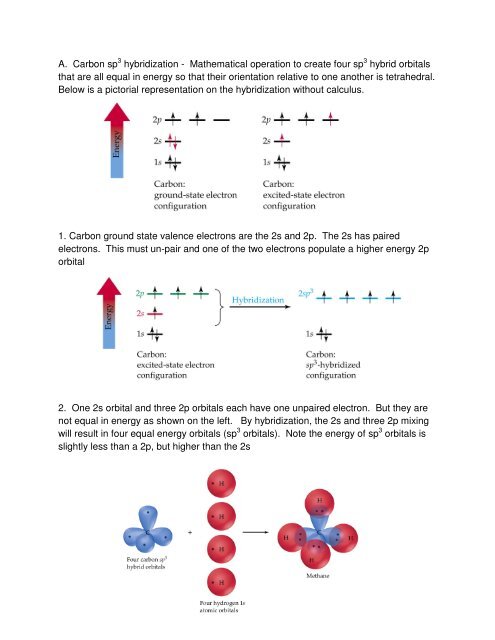
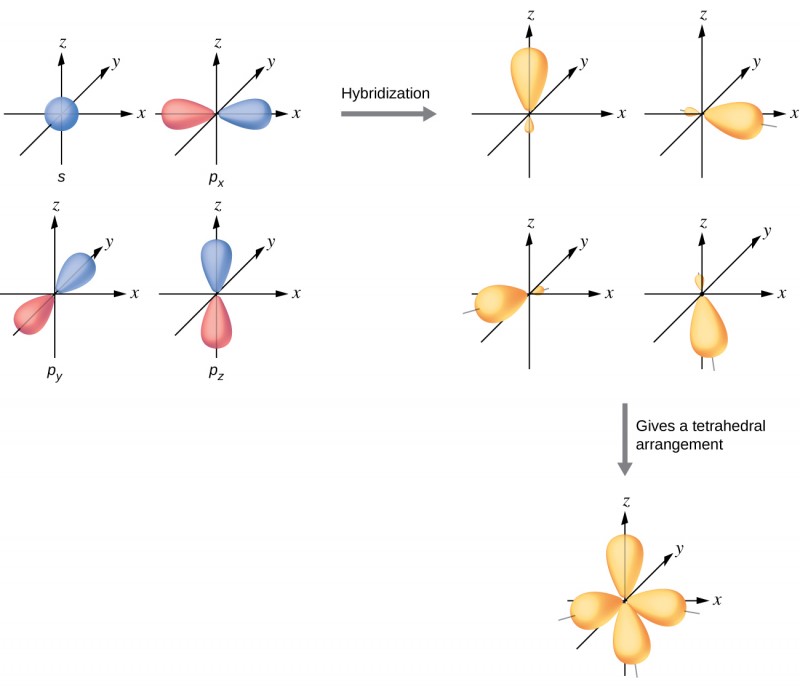
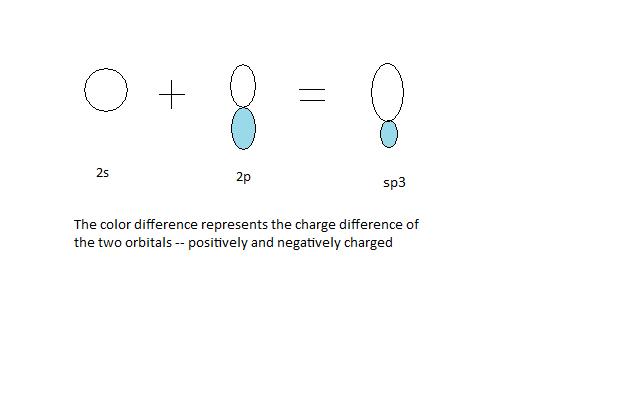
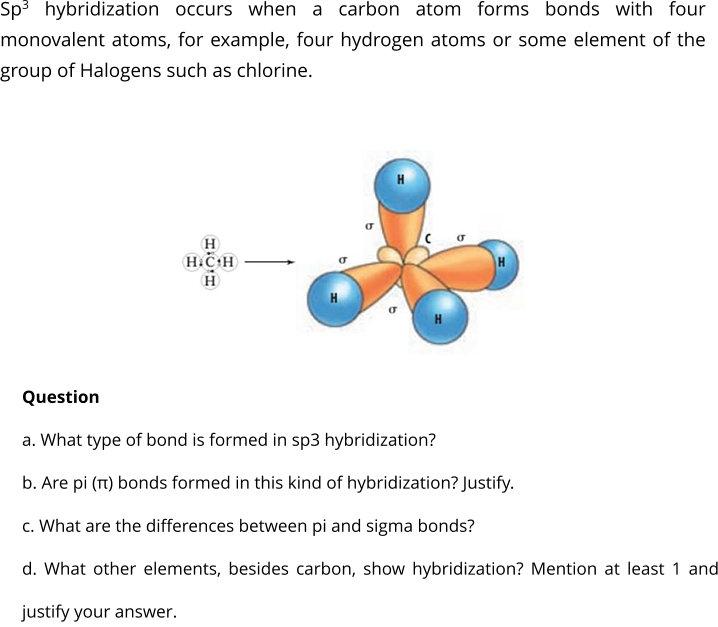
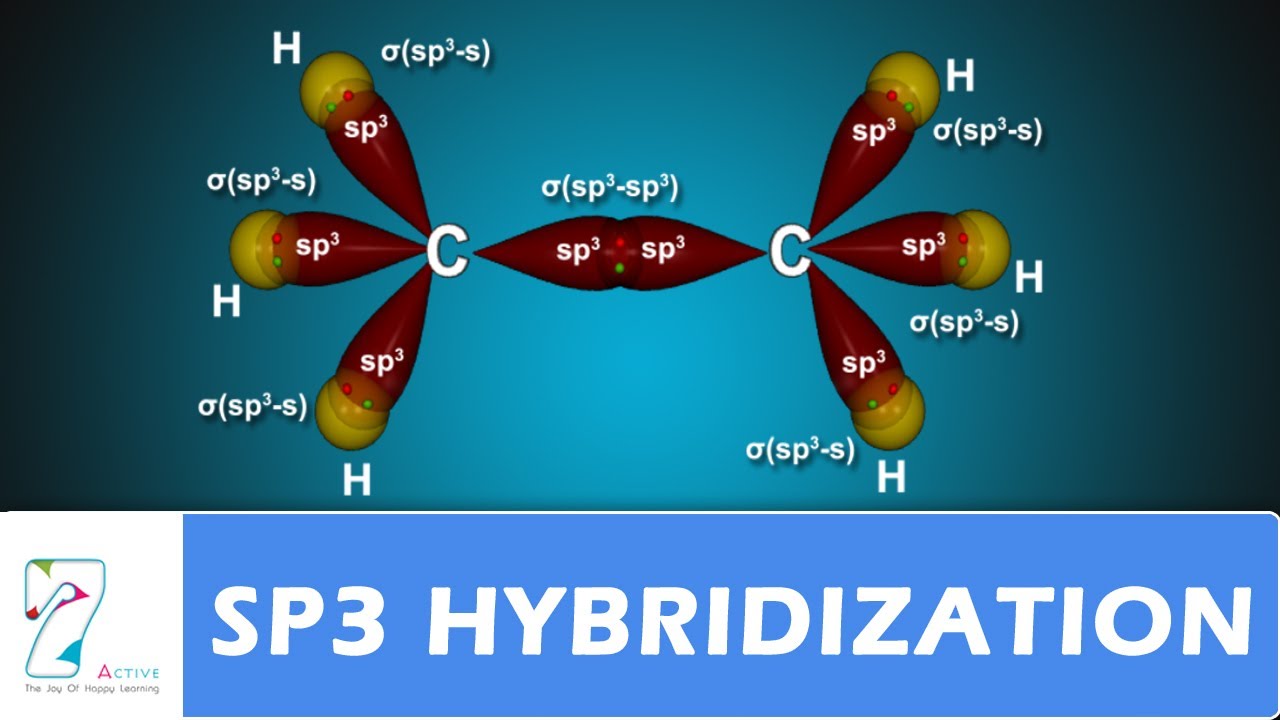
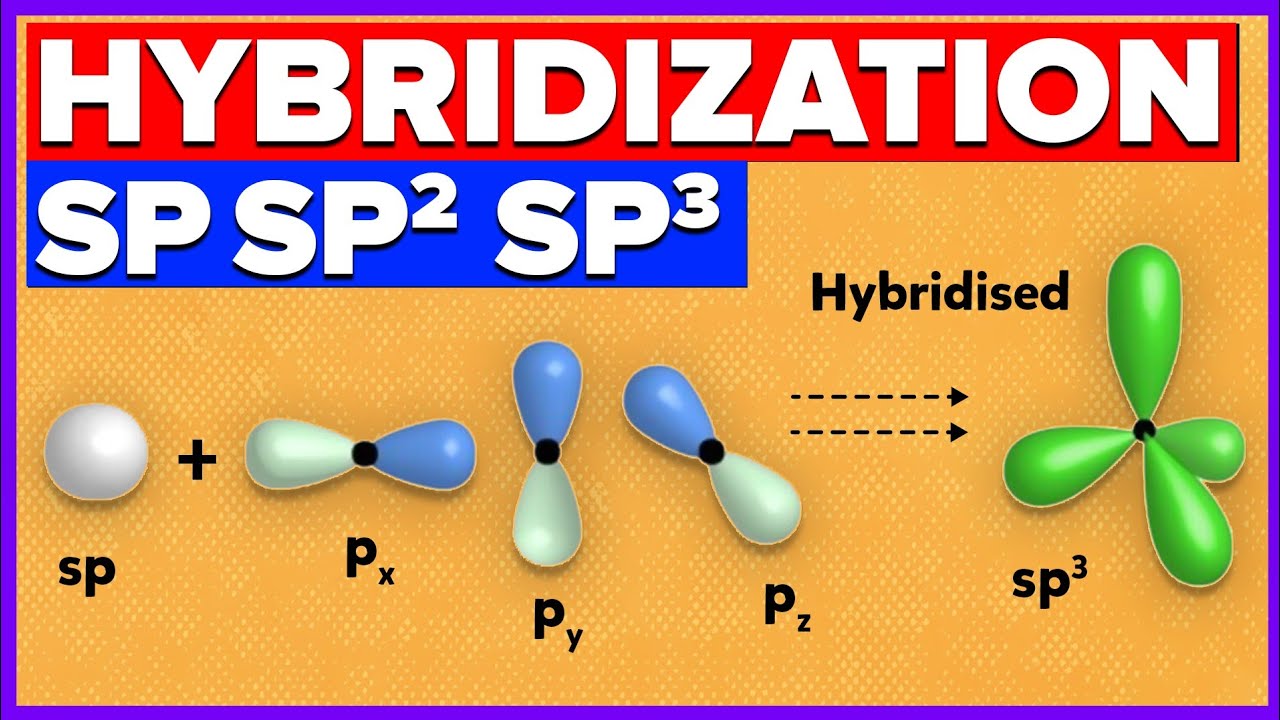
How does carbon hybridize its s and p orbitals and what effect does this have on the 3D structure and chemical behavior? Your answer must include diagrams of sp, sp2, and sp3

Monitoring the thermally induced transition from sp3-hybridized into sp2- hybridized carbons - ScienceDirect

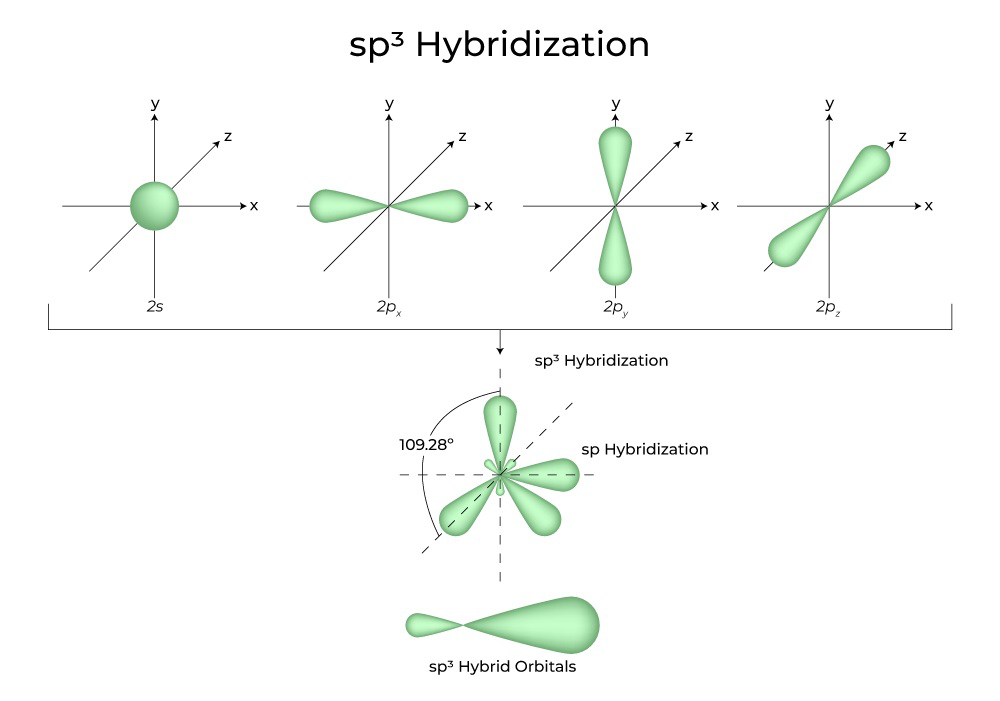
/chapter2/pages1and2/page1and2_files/sp2hyb.png)