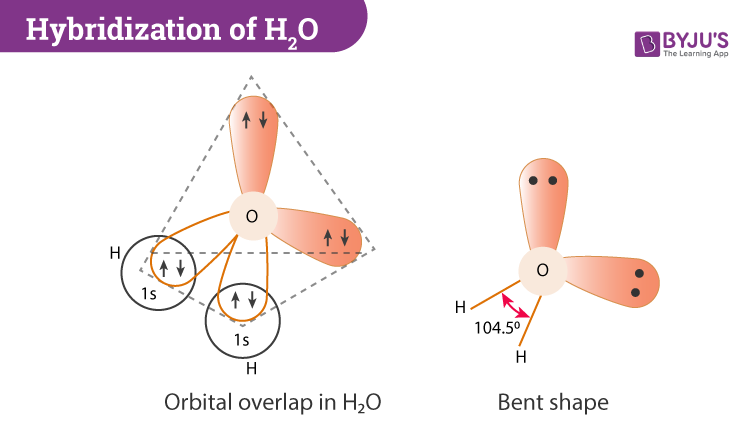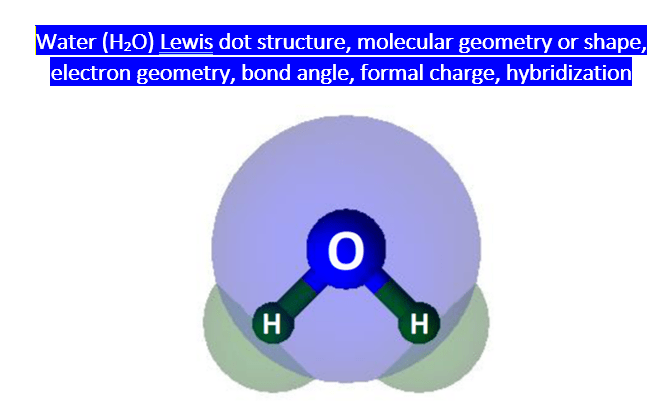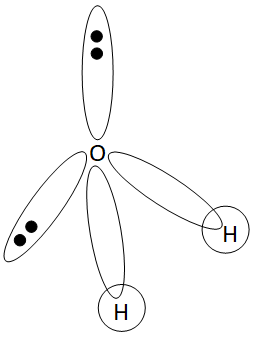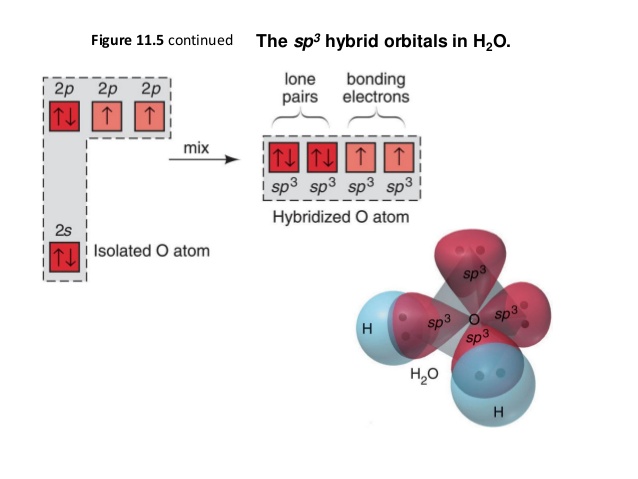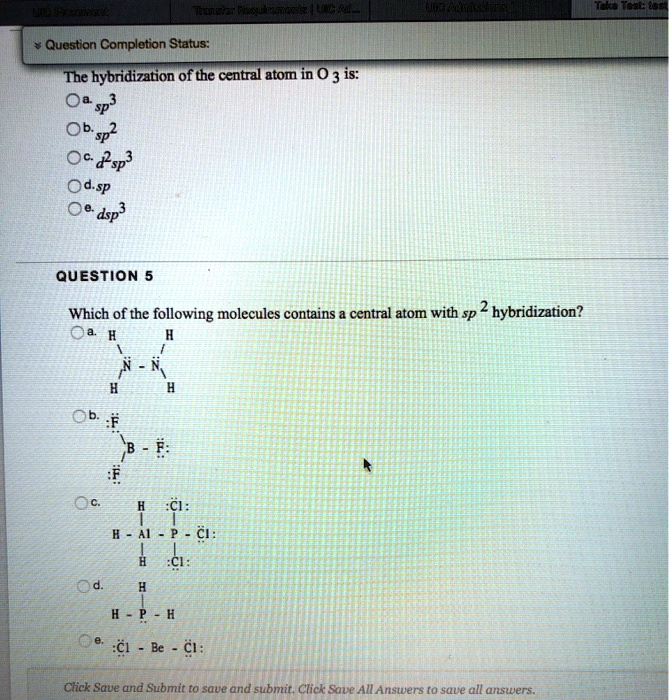
SOLVED: Q4L hybridization of the central atom in O3 is: dsp Which of the following molecules contains a central atom with sp2 hybridization? H2O Cl2 CO2
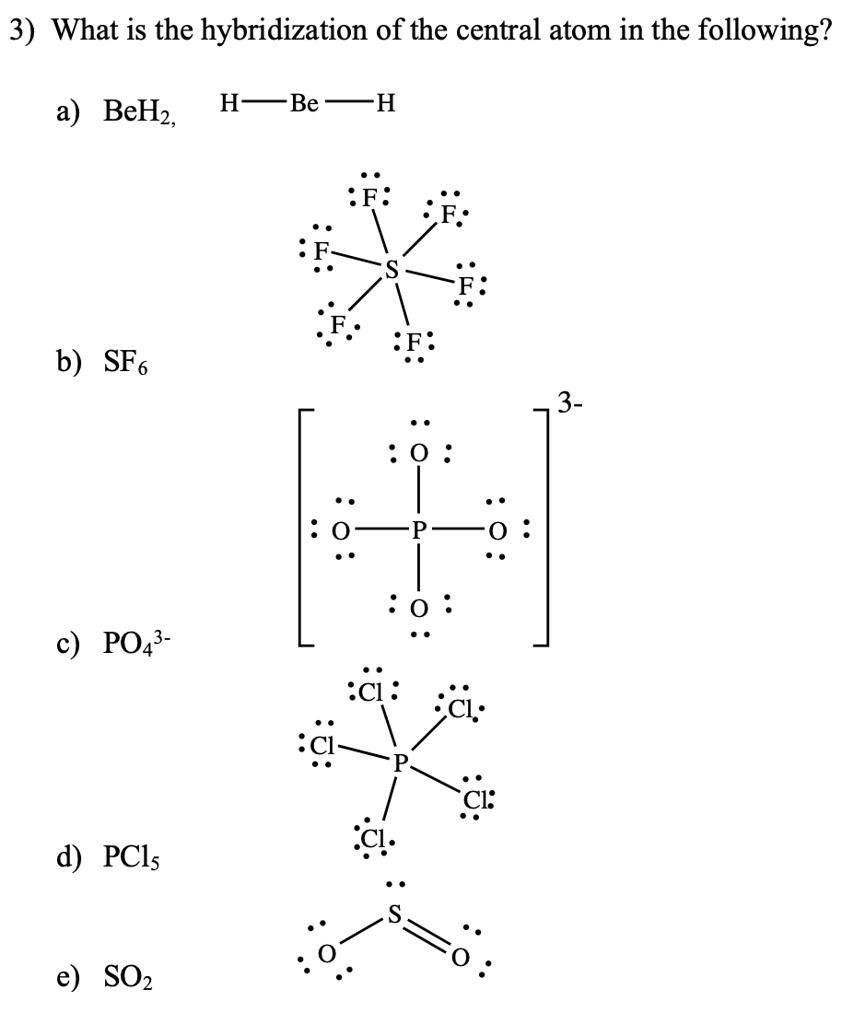
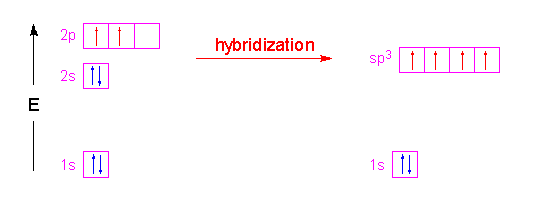
SOLVED: What is the hybridization of the central atom in the following? H2O a) BeH2, b) SF6 c) PO3^- d) PCl5 e) SO2

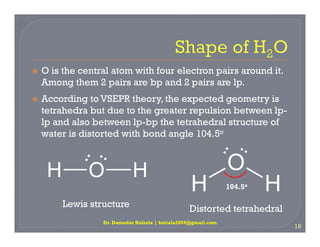
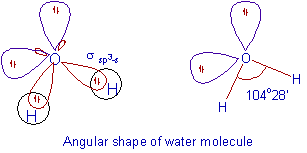
Chemistry - Molecular Structure (40 of 45) Hybridization with 2 Free Electron Pairs - Water - H2O - YouTube
![Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE](https://edurev.gumlet.io/ApplicationImages/Temp/634ddd4f-b583-4cd5-93a8-2591942ac108_lg.gif?w=360&dpr=2.6)
Hybridisation of the underline atom changes in: [2002]a)AlH3 changes to AlH 4-b)H2O changes to H3O+c)NH3 changes to NH4+d)in all casesCorrect answer is option 'A'. Can you explain this answer? - EduRev JEE