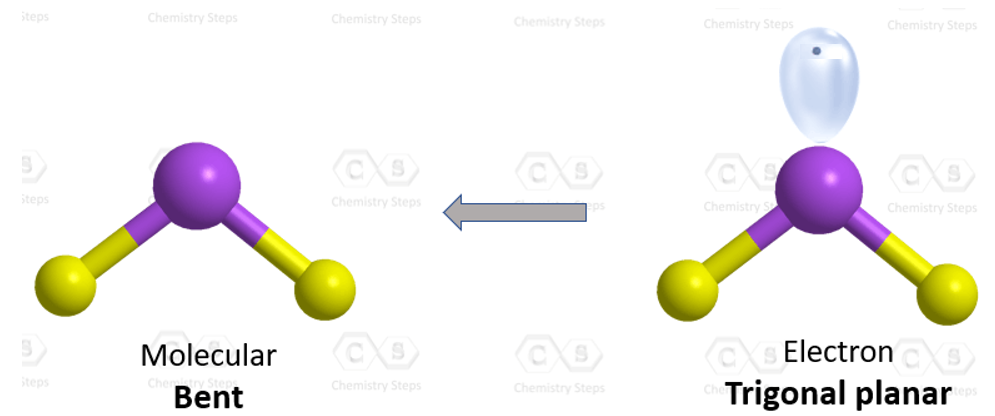
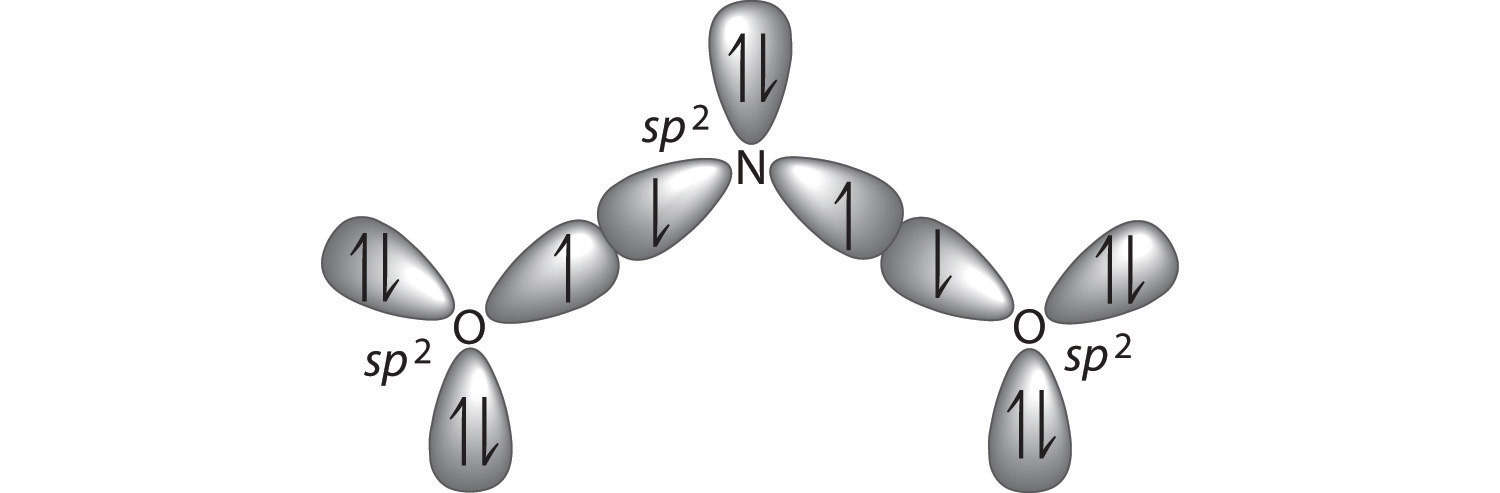
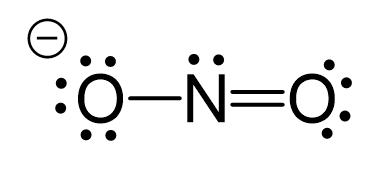The hybridization of atomic orbitals of nitrogen is `NO_(2)^(+), NO_(3)^(-)`, and `NH_(4)^(+)` respe - YouTube
The hybridisation of orbitals of N atom in NO3^–, NO2^+ and NH4^+ are respectively - Sarthaks eConnect | Largest Online Education Community
The hybridization of orbitals of N atom in NO3^- NO2^+ and NH4^+ are respectively - Sarthaks eConnect | Largest Online Education Community
The hybridization of atomic orbitals of nitrogen in NO2^+ , NO^-2 and NH4^+ are - Sarthaks eConnect | Largest Online Education Community

Bond Angle And Hybridization Of NO2,NO2-,NO2+,NO3-||Lewis Dot Structure||iit,neet,cbse, icse,kvpy|| - YouTube